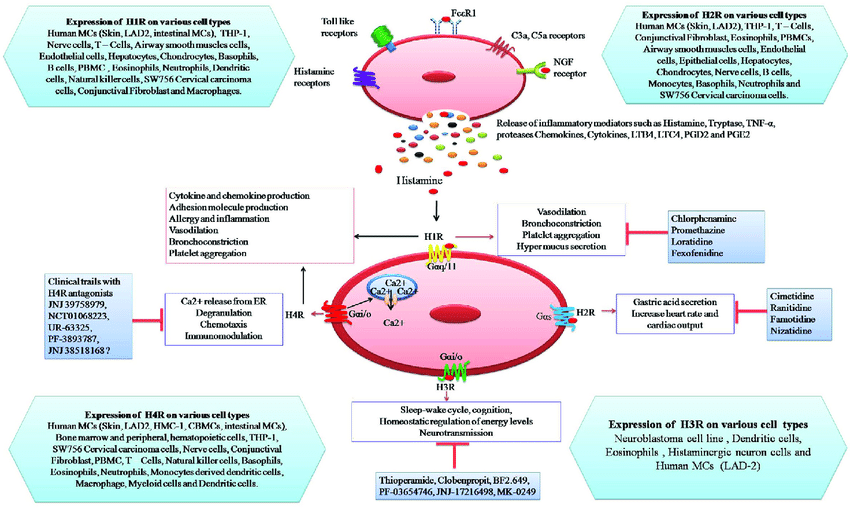
Conditional Expression Of A Gi
See allHide authors and affiliations
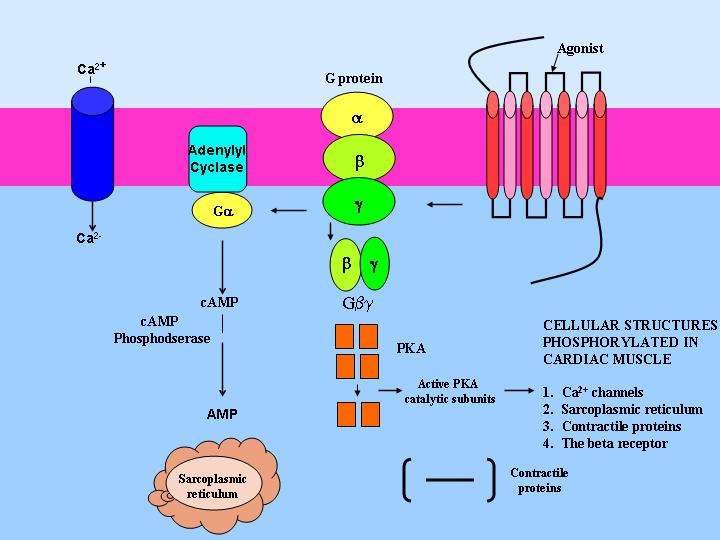
Correlation Between Heart Rates And Cardiac Output
Initially, physiological conditions that cause HR to increase also trigger an increase in SV. During exercise, the rate of blood returning to the heart increases. However as the HR rises, there is less time spent in diastole and consequently less time for the ventricles to fill with blood. Even though there is less filling time, SV will initially remain high. However, as HR continues to increase, SV gradually decreases due to decreased filling time. CO will initially stabilize as the increasing HR compensates for the decreasing SV, but at very high rates, CO will eventually decrease as increasing rates are no longer able to compensate for the decreasing SV. Consider this phenomenon in a healthy young individual. Initially, as HR increases from resting to approximately 120 bpm, CO will rise. As HR increases from 120 to 160 bpm, CO remains stable, since the increase in rate is offset by decreasing ventricular filling time and, consequently, SV. As HR continues to rise above 160 bpm, CO actually decreases as SV falls faster than HR increases. So although aerobic exercises are critical to maintain the health of the heart, individuals are cautioned to monitor their HR to ensure they stay within the target heart rate range of between 120 and 160 bpm, so CO is maintained. The target HR is loosely defined as the range in which both the heart and lungs receive the maximum benefit from the aerobic workout and is dependent upon age.
Other Factors Influencing Heart Rate
Using a combination of autorhythmicity and innervation, the cardiovascular center is able to provide relatively precise control over HR. However, there are a number of other factors that have an impact on HR as well, including epinephrine, NE, and thyroid hormones; levels of various ions including calcium, potassium, and sodium; body temperature; hypoxia; and pH balance. Table 1 and Table 2;illustrate the effect each factor has on heart rate. ;After reading this section, the importance of maintaining homeostasis should become even more apparent.
| Table 1. Major Factors Increasing Heart Rate and Force of Contraction |
|---|
| Factor |
Also Check: Does Acid Reflux Cause Heart Palpitations
A Mouse Model Of Inducible Cardiomyopathy
The Ro1 model of cardiomyopathy provides several insights that may advance our understanding of the etiology of cardiomyopathy. First, it demonstrates that the expression of a Gi-coupled receptor in mouse heart can cause a lethal cardiomyopathy with systolic dysfunction and LV chamber dilation. In contrast to previous data suggesting that up-regulation of the Gi pathway in cardiomyopathy is a compensatory response to hyperactive Gs signaling, our findings suggest a causal role for the Gi pathway. Second, our PTX experiments prove that Gi signaling causes ventricular conduction delay, a cardinal feature of dilated cardiomyopathy associated with a poor prognosis . The cause of ventricular conduction delay in human cardiomyopathy is unknown. Our findings raise the possibility that Gi signaling plays a similar role in humans. Third, the Ro1 model allows precise timing of both gene induction and gene suppression in an adult mouse, which should allow dissection of the steps leading to this cardiomyopathy and those leading to recovery.
Factors Decreasing Heart Rate
HR can be slowed when a person experiences altered sodium and potassium levels, hypoxia, acidosis, alkalosis, and hypothermia.;The relationship between electrolytes and HR is complex, but maintaining electrolyte balance is critical to the normal wave of depolarization. Of the two ions, potassium has the greater clinical significance. Initially, both hyponatremia and hypernatremia may lead to tachycardia. Severely high hypernatremia may lead to fibrillation, which may cause CO to cease. Severe hyponatremia leads to both bradycardia and other arrhythmias. Hypokalemia also leads to arrhythmias, whereas hyperkalemia causes the heart to become weak and flaccid, and ultimately to fail.
Heart muscle relies exclusively on aerobic metabolism for energy. Hypoxia leads to decreasing HRs, since metabolic reactions fueling heart contraction are restricted.
Don’t Miss: Does Acid Reflux Cause Heart Palpitations
Cardiac Sympathetic Nervous System
The sympathetic nervous system is the component of the ANS that is responsible for controlling the human bodys reaction to situations of stress or emergency , while the parasympathetic nervous system is generally responsible for basal organ system function.
Cardiac sympathetic preganglionic nerves emerge from the upper thoracic segments of the spinal cord . After traveling a short distance, preganglionic fibers leave the spinal nerves through branches called white rami and enter sympathetic ganglia. The cardiac sympathetic neurons form the sympathetic chain ganglia located along the side of the viscera column . These ganglia comprise the sympatheric trunks with their connecting fibers. The postganglionic fibers, extend to the viscera, such as the heart. In general, sympathetic preganglionic neurons are shorter than sympathetic postganglionic neurons .
Sympathetic receptors: There are two types of adrenergic receptors: and . In the cardiovascular system there are 1, 2, 1, and 2 adrenergic receptors .
Heart Rate And Heart Rate Variability In Posttraumatic Disorder
Increased heart rate is a determinant of physiological arousal seen in PTSD. Subjects who develop PTSD have been shown to have higher heart rates during the immediate aftermath of the trauma compared to traumatized individuals who do not develop PTSD. The elevated heart rate is caused by the increased noradrenergic tone, suggesting that increased noradrenergic activity immediately after the trauma may play an important role in the neurobiological processes involved in the development of PTSD. From a clinical perspective, this finding suggests that elevated heart rate immediately after the trauma is a predictor of PTSD.
Michael J. Aminoff, in, 2012
Don’t Miss: Does Tylenol Increase Heart Rate
Suppression Of Ro1 Expression Partially Reverses Cardiomyopathy
We predicted from our survival curve data that suppressing Ro1 expression after 8 wk would reverse the cardiac pathologic and physiologic changes. To test this hypothesis, we suppressed Ro1 expression for 4 wk in the same nine mice that underwent echocardiographic examination after 8 wk of Ro1 expression. In mice with signs of clinical distress, suppression of Ro1 caused rapid improvement in breathing, activity, and fluid balance . Pathologic examination of the hearts of these mice revealed no reduction in collagen deposition as compared with the 8-wk time point . By echocardiography, suppression of Ro1 expression significantly improved aortic peak flow velocity and LVFS . These results demonstrate that suppressing Ro1 expression prevents further mortality and allows partial recovery of cardiac function, but does not reverse some of the structural changes, such as chamber dilation and collagen deposition .
Iiiryr2 In Cardiac Contraction And Pacemaking
The cardiac action potential triggers DHPRs causing a rise in C and activation of RyR2 channels in the SR via their cytoplasmic facing Ca2+-activation sites. The subsequent release of Ca2+ from the SR further increases C by feeding back to cause further RyR2 activation. This process, known as Ca2+-induced Ca2+ release , provides a strong positive feedback to open more RyR2. In this way, Ca2+ release from the SR contributes up to 95% of the Ca2+ entering the cytoplasm during EC coupling . Shortly after this, the positive feedback cycle of CICR is broken due to the large resultant decrease of Ca2+ in the SR lumen which causes RyR2 to close and hence SR Ca2+ release ceases. The excess Ca2+ in the cytoplasm is either pumped back into the SR by ATP-driven Ca2+ pumps in the SR membrane or extruded from the cell by the Na/Ca exchanger in the sarcolemma during diastole. The major Ca2+ fluxes are Ca2+ uptake and release from the SR by SERCA2a and RyR2 and uptake and release from the cell by DHPRs and the NCX . These mechanisms serve a fundamental role in the large changes in free in the cytoplasm and SR lumen between diastole and systole, respectively .
D. Wu, J. Cui, in, 2014
Also Check: How To Calculate Target Heart Rate Zone
Does Increased Gi Signaling Cause Cardiomyopathy In Humans
Gi-coupled receptors in the heart include receptors for opiates , endothelin, somatostatin, and acetylcholine , as well as many orphan receptors identified by the Human Genome Project. These Gi-coupled receptors could cause cardiomyopathy by several mechanisms. First, activating mutations of GPCRs could increase basal signaling levels enough to cause cardiac disease. Similar mutations have been shown to cause human thyroid hyperplasia, premature testicular maturation, and other endocrinopathies . Second, increased levels of ligands acting on any of these receptors could activate Gi signaling and contribute to cardiomyopathy. Third, autoantibodies that bind to and cause signaling through endogenous Gi-coupled receptors could cause increased Gi signaling in the heart , as occurs in inflammatory insults to myocardium, such as Chagas cardiomyopathy. In each of these disease models, chronic activation of the Gi pathway would result in a phenotype similar to that in the Ro1 model. By showing that the Gi pathway has a possible causative role in prolonged ventricular conduction and cardiomyopathy, we implicate an entire class of GPCRs and provide insights into a wide variety of potential therapeutic targets for this syndrome.
Determinants Of Action Potential Generation
After an AP is triggered, the cardiac myocyte is unable to initiate another AP for some duration of time . This period of time is referred to as the refractory period. The refractory period is usually subdivided into an absolute refractory period, the interval during which a second AP absolutely cannot be initiated no matter how large a stimulus is applied, and relative refractory period, an interval immediately following the absolute refractory period during which initiation of a second AP is inhibited but not impossible. The myocyte is absolutely refractory when a critical mass of Na+ channels is unavailable for the all-or-nothing response . Na+ channels remain inactivated after activation and during depolarization until the membrane hyperpolarizes, at which point they transition to a closed state and undergo de-inactivation, regaining the ability to open in response to a stimulus. In the relative refractory period, enough Na+ channels have recovered from inactivation but K+ conductance of the membrane is sufficiently high that a large magnitude stimulus is required in order to reach the initiation threshold for a second AP.
Derek R. Laver, in, 2010
Read Also: How Do You Calculate Max Heart Rate
Chronic Expression Of Ro1 Causes Pathologic And Physiologic Changes Characteristic Of Cardiomyopathy
All 14 mice that died after long-term Ro1 expression had clinical or necropsy evidence of a cardiomyopathy. Just before death, 8 of the 14 mice developed signs of clinical distress characterized by labored breathing, rough hair coat, complete inactivity, failure to eat or drink, and fluid retention . At necropsy, these 8 mice had edema, ascites, and pleural effusions. Other mice showed signs of dehydration caused by lack of fluid intake . Cross sections of hearts from all 14 mice demonstrated enlarged ventricular chamber size and decreased ventricular wall thickness compared with controls .
Expression of Ro1 in mouse heart causes a cardiomyopathy. Control mouse and Ro1-expressing mouse with anasarca . Photomicrographs of hematoxylin/eosin-stained cross sections of mouse heart. The ventricular chambers were larger and the ventricular walls thinner in the mouse expressing Ro1 than in the control mouse .
Myocardial force was measured in right ventricular papillary muscle from mice expressing Ro1 . Absolute force was 50% lower in mice expressing Ro1 than in control mice . Furthermore, the rates of muscle contraction and relaxation were prolonged in Ro1-expressing mice . These results suggest that expression of a Gi-coupled receptor for 8 wk in mouse myocardium impairs both the absolute force generated by the heart and the time course of contraction and relaxation. These physiologic findings are consistent with the observed phenotype of cardiomyopathy.
Sympathetic Nervous System Receptors
The types of sympathetic or adrenergic receptors are alpha, beta-1;and beta-2. Alpha-receptors are located on the arteries. When the alpha receptor is stimulated by epinephrine or norepinephrine, the arteries constrict. This increases the blood pressure and the blood flow returning to the heart. The blood vessels in skeletal muscles lack alpha-receptors because they need to stay open to utilize the increased blood pumped by the heart.
Also Check: Can Anemia Cause Heart Failure
Health And Performance Considerations
Higher heart rates may be an indication of poor heart function and higher than usual stress being placed on the hearts ability to circulate blood. This may further indicate heart disease conditions.
From a performance stand point knowing specific heart rate training zones can optimize our bodys ability to adapt to performance requirements. Determining these zones can be done through many different methods, including VO2 or lactate testing, formulas and general training regimens. It then becomes necessary to monitor intensity in order to optimize your chances for success. To monitor your intensity there are several methods available to you. First is the perceived exertion method in which you rate your perception of how hard you are exerting yourself during a workout. The acronym for this is RPE . The scale on which to base your perceptions range from 1 – 10. See below.
The scale can be broken down as follows:
;;;;;0: Nothing
Disorders Of The Heart: Abnormal Heart Rates
For an adult, normal resting HR will be in the range of 60100 bpm. Bradycardia is the condition in which resting rate drops below 60 bpm, and tachycardia is the condition in which the resting rate is above 100 bpm. Trained athletes typically have very low HRs. If the patient is not exhibiting other symptoms, such as weakness, fatigue, dizziness, fainting, chest discomfort, palpitations, or respiratory distress, bradycardia is not considered clinically significant. However, if any of these symptoms are present, they may indicate that the heart is not providing sufficient oxygenated blood to the tissues. The term relative bradycardia may be used with a patient who has a HR in the normal range but is still suffering from these symptoms. Most patients remain asymptomatic as long as the HR remains above 50 bpm.
Read Also: Tylenol Heart Palpitations
What Controls Heart Rate
Heart rate is controlled by the two branches of the autonomic nervous system. The sympathetic nervous system and the parasympathetic nervous system . The sympathetic nervous system releases the hormones to accelerate the heart rate. The parasympathetic nervous system releases the hormone acetylcholine to slow the heart rate. Such factors as stress, caffeine, and excitement may temporarily accelerate your heart rate, while meditating or taking slow, deep breaths may help to slow your heart rate.;; Exercising for any duration will increase your heart rate and will remain elevated for as long as the exercise is continued. At the beginning of exercise, your body removes the parasympathetic stimulation, which enables the heart rate to gradually increase. As you exercise more strenuously, the sympathetic system kicks in to accelerate your heart rate even more. Regular participation in cardiovascular exercise over an extended period of time can decrease your resting heart rate by increasing the hearts size, the contractile strength and the length of time the heart fills with blood. The reduced heart rate results from an increase in activity of the parasympathetic nervous system, and perhaps from a decrease in activity of the sympathetic nervous system.
Exercise And Maximum Cardiac Output
In healthy young individuals, HR may increase to 150 bpm during exercise. SV can also increase from 70 to approximately 130 mL due to increased strength of contraction. This would increase CO to approximately 19.5 L/min, 45 times the resting rate. Top cardiovascular athletes can achieve even higher levels. At their peak performance, they may increase resting CO by 78 times.
Since the heart is a muscle, exercising it increases its efficiency. The difference between maximum and resting CO is known as the cardiac reserve. It measures the residual capacity of the heart to pump blood.
You May Like: How Much Blood Does The Heart Pump
Disorders Of The Heart: Broken Heart Syndrome
Extreme stress from such life events as the death of a loved one, an emotional break up, loss of income, or foreclosure of a home may lead to a condition commonly referred to as broken heart syndrome. This condition may also be called Takotsubo cardiomyopathy, transient apical ballooning syndrome, apical ballooning cardiomyopathy, stress-induced cardiomyopathy, Gebrochenes-Herz syndrome, and stress cardiomyopathy.
The recognized effects on the heart include congestive heart failure due to a profound weakening of the myocardium not related to lack of oxygen. This may lead to acute heart failure, lethal arrhythmias, or even the rupture of a ventricle. The exact etiology is not known, but several factors have been suggested, including transient vasospasm, dysfunction of the cardiac capillaries, or thickening of the myocardiumparticularly in the left ventriclethat may lead to the critical circulation of blood to this region. While many patients survive the initial acute event with treatment to restore normal function, there is a strong correlation with death. Careful statistical analysis by the Cass Business School, a prestigious institution located in London, published in 2008, revealed that within one year of the death of a loved one, women are more than twice as likely to die and males are six times as likely to die as would otherwise be expected.
Effects Of Temperature Changes
The cardiac AP is strongly changed by temperature . Lowering of temperature increases the duration and reduces the rate of upstroke of cardiac AP. As a consequence, heart rate and velocity of impulse conduction over the heart are depressed, and the duration of contraction of each cardiac myocyte lasts longer. The longer duration of AP allows more time for calcium influx through the sarcolemma and at least partly compensates for the temperature-dependent decrease in calcium influx via ICa. Prolonged depolarization also allows more time for the myofilaments to generate force.
Fig. 4. Temperature has strong effect on the duration of fish cardiac AP and contraction. Continuous recording of AP from a ventricular myocyte of crucian carp when temperature was gradually elevated from 5 to 18°C. Note the shortening of AP duration and increase in RMP. Effects of temperature change on ventricular AP and associated contractions in crucian carp heart at 4 and 18°C.
H.A. Shiels, in, 2011
Don’t Miss: How Accurate Is Fitbit Charge 2 Heart Rate
