Second Stage Of Treatment
Currently, two basic surgical techniques are used: the hemi-Fontan operation or bidirectional Glenn anastomosis. The energy loss in the lateral intra-atrial tunnel produced after the previous hemi-Fontan operation is significantly lower compared to the energy dissipation in the tunnel produced after Glenns two-way operation. More physiological distribution of blood to both lungs after the hemi-Fontan surgery has been also noted . The second stage of treatment after previous treatment hybridization is a more complicated and longer operation. It is a combination of the Norwood operation and hemi-Fontan operation and in some centers also serves as preparation for performing the third stage of invasive cardiological defect treatment .
Traditionally, the bidirectional Glenn anastomosis was situated between the superior vena cava and the pulmonary arteries. The inferior vena cava to pulmonary artery anastomosis was abandoned in the animal lab by Dr. Glenn after repeated failures in his animal models. In patients with unfavorable upper body systemic venous anatomy, the superior vena cava –pulmonary artery connection is suboptimal or not feasible, and an alternative is needed to unload the heart. As it was demonstrated by studies, this subset of patients may benefit from the primary inferior vena cava -pulmonary artery connection, the Southern Glenn, which we have performed successfully in two patients .
Our Areas Of Innovation For Hlhs
The Boston Childrens Benderson Family Heart Center offers a unique combination of technical expertise, a state-of-the-art cardiac intensive care unit, and a continuous drive towards innovation that allows us to help when others cannot. We are here to serve as a resource to you. Some of our innovations include:
- The stage 1 operation was initially developed and first performed at Boston Childrens Hospital.
- We were the first center to perform a successful fetal cardiac intervention and have done more fetal interventions for babies with HLHS than any other center in the world.
- In our Congenital Heart Valve Program, we have pioneered surgical techniques for repairing the tricuspid valve in babies with HLHS and tricuspid regurgitation.
- In our Complex Biventricular Repair Program, we have learned how to recruit the smaller ventricles to make two pumping chambers instead of one.
Medical Treatment After Birth
If fetal intervention is not an option for your baby, treatments after birth will focus on stabilizing your baby and may include:
- medication to keep the patent ductus arteriosus open
- intravenous fluids through a tube inserted into a vein
- a feeding tube if your baby has difficulty feeding
- breathing assistance provided by a ventilator to ensure adequate oxygen
- a balloon atrial septostomy procedure to create an opening in the wall between the upper chambers of the heart to improve mixing of oxygen-rich blood and oxygen-poor blood
Shortly after birth, your childs care team will take images of your babys heart using an echocardiogram to see every detail of your babys heart the way the blood flows, the size and function of the ventricles, and how well the valves work.
Recommended Reading: Heart Attack In Man
How We Care For Hlhs
At Boston Childrens Hospital, we have a team of specialists ready to evaluate your baby starting in the Maternal Fetal Care Center. This team includes cardiologists who specialize in fetal assessments, and a team of nurses and other specialists who will make sure your baby is in the best possible condition when born. They will help you plan for every step along the way. In some cases, we are able to offer a fetal cardiac intervention even before the baby is born.
Boston Childrens heart surgeons treat a high volume of some of the most complex pediatric heart conditions in the world, with survival rates of more than 98 percent. Our approach to treating HLHS is different for every child, based upon their individual needs. We are one of the few hospitals in the country to offer complex biventricular repair, an approach pioneered at Boston Childrens.
Between The Norwood And Glenn Operations
Though early outcomes for patients with single ventricle heart defects after staged reconstruction have improved dramatically, the period between the Norwood procedure and the Glenn operation remains a very vulnerable time for infants. CHOP created the Infant Single Ventricle Monitoring Program to focus on the care and monitoring of infants with single ventricle heart defects, such as HLHS, between the first and second reconstructive surgeries.
Don’t Miss: Congestive Heart Failure Stage 4
Is Hypoplastic Left Heart Syndrome Treatable
Yes. First, your baby will need a medication called prostaglandin. This keeps their ductus arteriosus open and functioning. Other medicines can help your babys heart work more efficiently. Your baby also may need help with breathing.
Then theyll need a series of surgeries to direct blood flow to their lungs and body. These operations put their hearts workload on their right ventricle, which does all the pumping.
Deterrence And Patient Education
Pregnant women who receive a fetal ultrasound consistent with a diagnosis of HLHS are referred to a pediatric cardiologist. Most pediatric cardiologists evaluate an expectant mother around 20 to 24 weeks gestation. The parents are given the treatment options and a frank discussion about the high morbidity and mortality associated with this congenital heart defect.
Read Also: Why Does Heart Rate Increase During Running
Treatment Option : Heart Transplant
Heart transplant involves replacing a sick heart with a new heart . There are risks involved with heart transplantation. Children will need to be on lifelong medications to prevent their own body from attacking the heart which is called rejection. Long-term complications of transplant include potential rejection, infection, coronary disease, and a special type of lymphoma associated with immunosuppression. Children who require the transplant will be placed on a waiting list administered by a national agency called United Network for Organ Sharing. The wait time can be days, months or even more than a year. Some babies may pass away while on the waiting list. A transplanted heart will last an average of 8-14 years. Some children also require repeat transplantation.
Hypoplastic Left Heart Syndrome Causes And Risk Factors
Some congenital heart defects may have a genetic link, either occurring due to a defect in a gene, a chromosome abnormality, or environmental exposure, causing heart problems to occur more often in certain families. In hypoplastic left heart syndrome, there may be abnormalities of other organs, such as diaphragmatic hernia, omphalocele, and hypospadias.
In many children, HLHS occurs sporadically , with no clear reason evident for their development.
Read Also: Congestive Heart Disease Symptoms
What Is Hypoplastic Left Heart Syndrome In Children
Hypoplastic left heart syndrome is a group of defects of the heart and large blood vessels. A child is born with this condition . It occurs when part of the heart doesn’t develop as it should during the first 8 weeks of pregnancy.
In hypoplastic left heart syndrome, most of the left side of the heart is small and underdeveloped. These structures are usually affected:
- Mitral valve. This valve controls blood flow between the left atrium and left ventricle.
- Left ventricle. This is the lower left chamber of the heart. It pumps blood to the body.
- Aortic valve. This valve controls blood flow from the left ventricle into the aorta, and then to the body.
- Aorta. This is the large artery leading from the left ventricle to the body.
The left ventricle is normally very strong so it can pump blood to the body. When it is small and poorly developed, it can’t pump any or enough blood out to the body. For this reason, an infant with hypoplastic left heart syndrome will not live long without surgery to fix the defect.
Treating Hypoplastic Left Heart Syndrome
Babies with HLHS need surgery in the first weeks of life. They get a series of surgeries to change blood flow through their heart.
Before surgery, your baby will need medicine that keeps the ductus arteriosus open so blood can get to the rest of their body. Your baby may need help breathing or need to be on a breathing machine .
HLHS is called a single-ventricle heart defect because there is only 1 pumping chamber in the heart, instead of the usual 2. Single-ventricle defects are some of the most complex heart birth defects. We provide comprehensive care for children with HLHS through our Single Ventricle Program.
Also Check: Which Part Of The Brain Controls Heart Rate
Variation In Care For Infants Undergoing The Stage Ii Palliation For Hypoplastic Left Heart Syndrome
2018, Cardiology in the Young
Abstract
BackgroundThe Single Ventricle Reconstruction trial randomised neonates with hypoplastic left heart syndrome to a systemic-to-pulmonary-artery shunt strategy. Patients received care according to usual institutional practice. We analysed practice variation at the Stage II surgery to attempt to identify areas for decreased variation and process control improvement.MethodsProspectively collected data were available in the Single Ventricle Reconstruction public-use database. Practice variation across 14 centres was described for 397 patients who underwent Stage II surgery. Data are centre-level specific and reported as interquartile ranges across all centres, unless otherwise specified.ResultsPreoperative Stage II median age and weight across centres were 5.4 months and 5.7 kg , with 70% performed electively. Most patients had pre-Stage-II cardiac catheterisation . Digoxin was used by 11/14 centres in 25% of patients , and 81% ha…
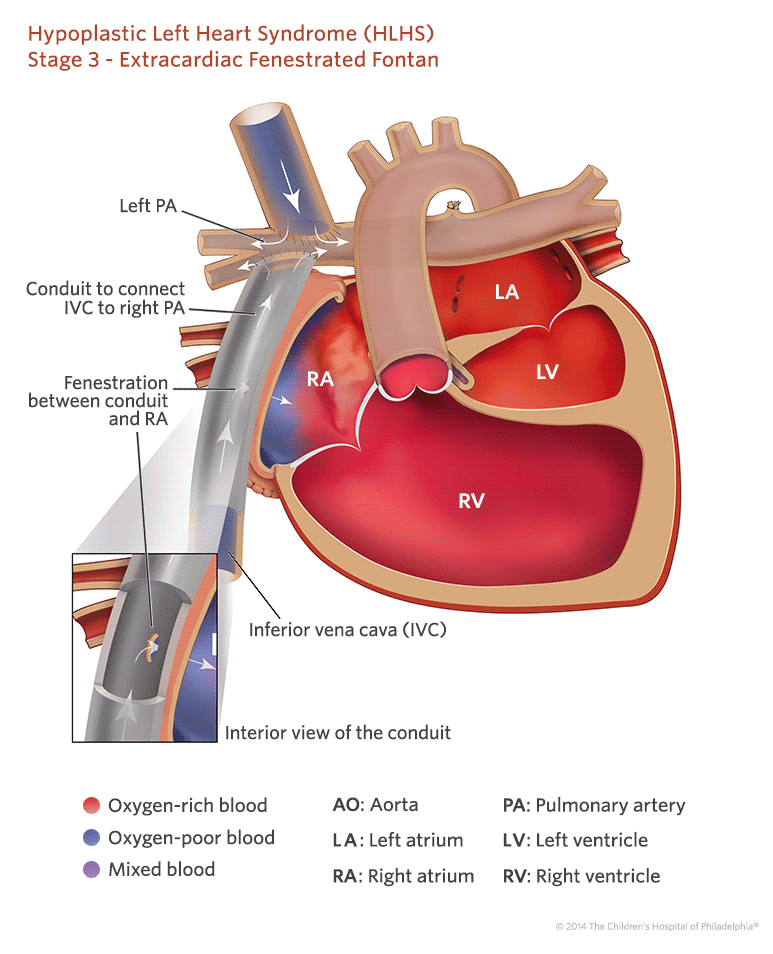
Stage Iii: Fontan Procedure
The final surgery usually takes place around 2 years of age. This stage requires open heart surgery in the Operating Room. The child is on the heart-lung machine the same as Stage II. At this time the blood from the lower part of the body that goes back to the heart through the inferior vena cava is routed to the pulmonary artery directly. This makes all of the blood returning from the body a more passive blood flow. After the final surgery is completed, the blood flow from the upper body will return to the lungs by the superior vena cava connection. Blood will reach the lungs from the lower body by the inferior vena cava connection.
Don’t Miss: Nursing Diagnosis For Congestive Heart Failure
Treatment For Hypoplastic Left Heart Syndrome
Hypoplastic left heart syndrome is most often fatal without early intervention. Compared to 25 years ago, there are now many different options for treatment of this complex heart condition an individualized approach is taken for each and every child. Your doctor will explain each individual option, and why one particular approach might be recommended for your child.
How Is Hypoplastic Left Heart Syndrome Treated
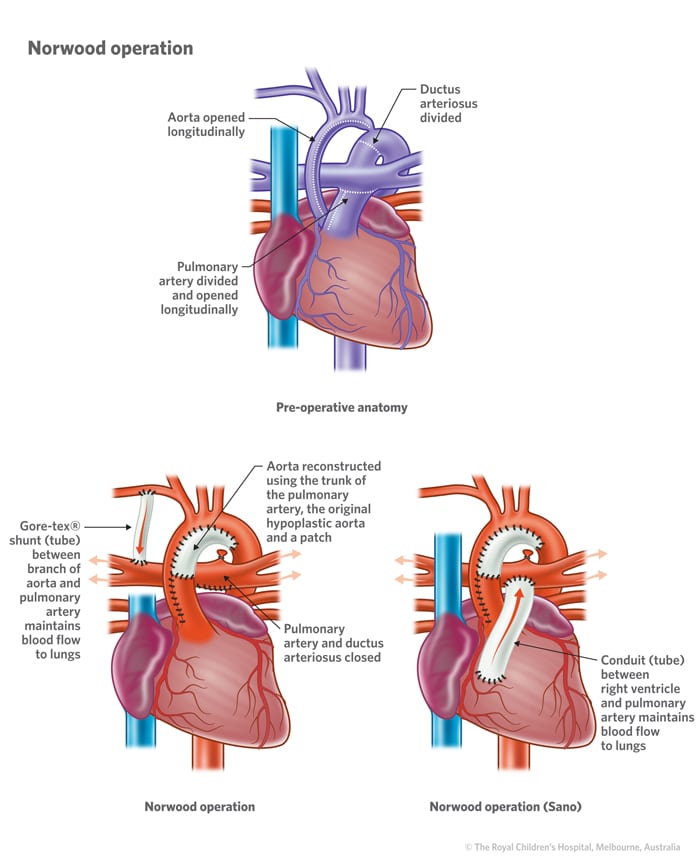
Surgeons perform three separate operations. Hypoplastic left heart syndrome surgery stages are the Norwood, Glenn and Fontan procedures. Your babys provider will need to do the surgeries in that order.
Norwood procedure: Babies with HLHS need Norwood surgery within the first two weeks of life. During the procedure, surgeons:
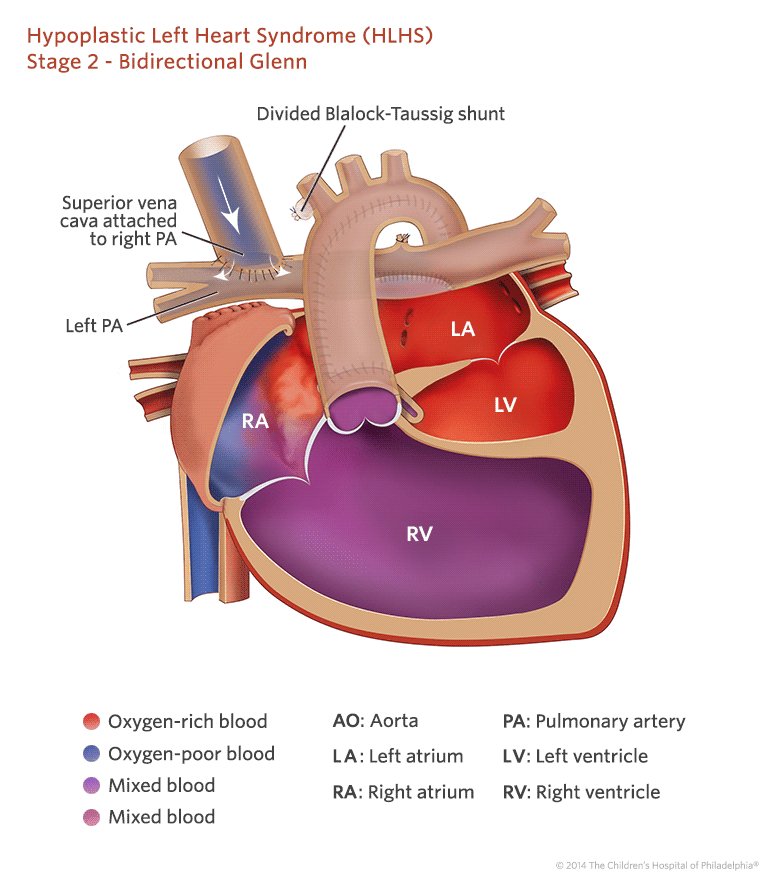
Bidirectional Glenn shunt operation: At 4 to 6 months of age, babies need a second operation. During the Glenn procedure, surgeons:
Complications of the treatment
Some children may not make it from one surgery to the next. Also, they may have problems after each surgery, such as:
- Issues with their right ventricle not working right.
- An aortic valve leak.
- Cardiac arrest.
Recommended Reading: Optical Heart Rate Monitor
Stage Ii And Stage Iii Procedures
Cardiovascular function was assessed by echocardiography and elective cardiac catheterisation before the bidirectional Glenn procedure and modified Fontan procedure . Thirty seven patients required cardiological intervention before stage II, primarily for neoaortic arch obstruction .25 Thirty six patients also required cardiological intervention before stage III. Most had balloon dilatation of the pulmonary arteries with or without stent insertion. Twelve patients had balloon dilatation of residual or previously untreated neoaortic arch obstruction.
Two hundred and three patients underwent a BDG at a median age of 4.9 months and median interval of 4.7 months after the Norwood procedure. Concomitant surgical procedures performed in 84 patients were pulmonary artery patch augmentation , patch augmentation of the neoaortic arch , atrioventricular valve annuloplasty , atrial septectomy , and insertion of a permanent pacemaker .
Hypoplastic Left Heart Syndrome Surgery
Typically, your child will require open heart surgery to re-direct the oxygen-rich blood and oxygen-poor blood. After these operations:
- The right side of the heart will do what is usually the job of the left side â pumping oxygenated blood to the body.
- The deoxygenated blood will flow from the veins to the lungs without passing through the heart.
The series of three reconstructive operations to repair HLHS â the Norwood, Glenn and Fontan procedures â is known as ” Staged Reconstruction .”
Frequent surveillance in infancy and early childhood is important to minimize risk factors for the eventual Fontan operation. Your child will also need a customized series of diagnostic tests between the planned stages of surgery, and throughout childhood. Additional surgical or catheter therapies, or in rare cases heart transplantation, may also be recommended.
Don’t Miss: What Blood Tests Detect Heart Problems
Stage : Fontan Procedure
This operation, called the Fontan procedure, occurs at approximately 1 1/2 to 3 years of age. During this surgery the inferior vena cava, a large vein that carries deoxygenated blood from the lower body into the heart, is disconnected from the heart and attached to the pulmonary artery. After this operation, all of the deoxygenated blood from the body goes to the lungs without passing through the heart.
Frequent surveillance in infancy and early childhood is important to minimize risk factors for the eventual Fontan operation. The Infant Single Ventricle Monitoring Program at CHOP was developed to provide care and monitoring for children between the first two surgical procedures.
Your child will also need a customized series of diagnostic tests between the planned stages of surgery, and throughout childhood. Additional surgical or catheter therapies, or in rare cases heart transplantation, may also be recommended.
Causes And Risk Factors
The causes of heart defects such as hypoplastic left heart syndrome among most babies are unknown. Some babies have heart defects because of changes in their genes or chromosomes. These types of heart defects also are thought to be caused by a combination of genes and other risk factors, such as things the mother comes in contact with in the environment or what the mother eats or drinks or the medicines the mother uses.
Hypoplastic left heart syndrome may be diagnosed during pregnancy or soon after the baby is born.
Teagan was born with hypoplastic left heart syndrome. Read his story as well as other stories from families affected by hypoplastic left heart syndrome »
Also Check: Foods To Avoid With Congestive Heart Failure
Caring For Your Child At Home Following A Hlhs Surgery
Pain medications, such as acetaminophen or ibuprofen, may be recommended to keep your child comfortable at home. Your child’s physician will discuss pain control before your child is discharged from the hospital.
If any special treatments are to be given at home, the nursing staff will ensure that you are able to provide them, or a home health agency may assist you.
You may receive additional instructions from your child’s physicians and the hospital staff. Families will also be referred to our Neurodevelopmental Follow-up Program, where our specialists will monitor and encourage your child’s healthy development for living life to the fullest.
Future Of Stem Cell Therapies
A systematic review found 23 articles, published since 2010, as well as nine relevant clinical trials related to congenital heart disease and recent advances in stem cell therapies. Pre-clinical research has focused on several types of stem cells including: mesenchymal stem cells , autologous umbilical cord blood cells, c-kit+ cardiac stem cells, and neonatal thymus mesenchymal stem cells. These cell types have shown the ability to differentiate into cardiac tissue making them ideal for cardiac regenerative therapy. The first use of autologous umbilical cord blood cells was done at the Mayo Clinic in 2015 and was found to increase right ventricular function in the patient after their procedure. The first use of cardiac progenitor cells occurred in the Transcoronary Infusion of Cardiac Progenitor Cells in Patients with Single-Ventricle Physiology Trail of 2011.
You May Like: Running Heart Rate Zones
What Happens After The Baby Is Born
Since the baby is connected to the mother by the placenta before birth, infants with HLHS usually do not have any problems before or during delivery. Every baby is born with a ductus arteriosus. This is a blood vessel connecting the pulmonary artery and the aorta and allows oxygen-rich blood from the placenta to get to the baby’s aorta. Usually, this blood vessel begins to close several hours to days after birth because the oxygen-rich blood now comes from the baby’s lungs and it is no longer needed. Because the left ventricle and aortic arch are small and underdeveloped in babies with HLHS, they need the ductus arteriosus open. If the ductus closes, blood flow to the body becomes very low and they can become very ill and even die. A special medicine called prostaglandin is used to keep the ductus arteriosus open after birth. The medicine is given through a vein until a procedure can be done to make a more permanent connection. One of the side effects of prostaglandin is apnea . If this occurs, it is sometimes necessary for a breathing tube to be placed and a breathing machine used to help the baby breathe.
